I have calcium crystals in my pool. What do I do?
Calcite crystals can sometimes be removed chemically, but it depends on what type of crystals you have.
Calcium crystals (or calcite crystals) can be a pain to get rid of. This article will briefly explain why they occur and how to fix the issue.

Why does my pool have calcium crystals?
Calcium crystals form on cementitious pool surfaces as a result of prolonged stagnant, aggressive water. Usually, they are formed during the winter when nobody is using the pool, and the pool is covered. Crystals are the result of low-LSI water stealing calcium from your pool surface. There are many things we still do not know about crystals, but our research is ongoing.
In fact, we know of at least four (4) different variations of crystals. Some are easy to remove, others are difficult to remove. But they're all preventable with proper winterization chemistry.
We think crystals are forming slowly as calcium is dissolved from a cementitious pool finish. Specifically, when calcium hydroxide is pulled from the cement into the water, its high pH (12.6) is high enough to precipitate calcium carbonate. But in cold, stagnant water, what appears to be happening is this calcium carbonate is building upon itself as it leaves the walls...much like a calcium nodule would. We believe this because the microscope views of these crystals show a tiny hollow channel in the middle of each crystal:
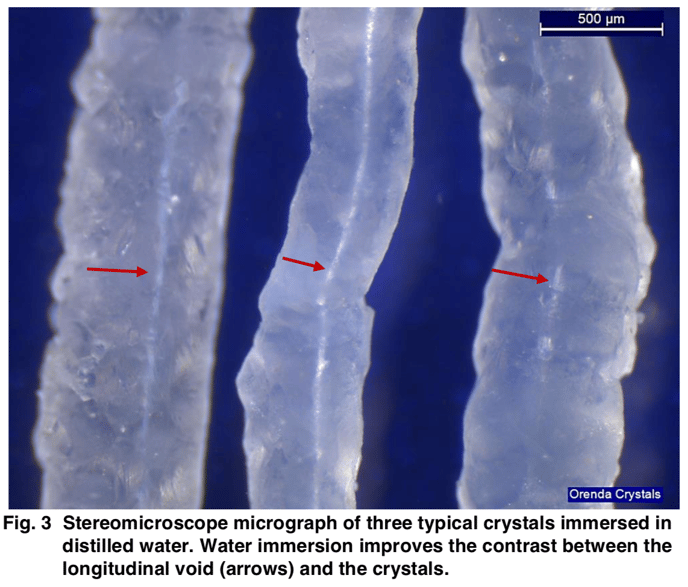
According to the lab, this indicates growth from the pool surface, NOT depositing from the water onto the surface. The lab described this as "skeletal calcite", which is not what carbonate scale looks like under a microscope.

How to clean up/remove calcium crystals in the pool
Thanks to lab testing samples, we know of at least four (4) types of calcite crystals. They're all similar, but for some reason, they form in different shapes, sizes, and densities. We don't know why. We do know that some are easier to remove than others.
The cleanup process is generally the same, though some can take longer than others. And still some crystals are so stubborn they just will not chemically dissolve. We have literally poured muriatic acid on a wall with crystals and it did nothing. But when we broke the crystals off, they fizzed and dissolved in the acid without an issue. It is the oddest thing and we have not found anyone who can explain it to us.
Anyway, try these steps, because these usually work for most pools with crystals:
- Grab a sample of water in a clean bottle. If the water is below 60ºF, warm up the sample before testing the water. Then test the water and input the results into the Orenda app on the left side (under "current levels").
- Adjust the calculator to reduce pH below 7.8, but increase calcium (most likely needed) up to 400+ ppm. If your water was cold enough to form crystals, your pool is in a climate cold enough to justify 350-400+ ppm calcium hardness at a minimum, but in our experience, aiming for 400-450 ppm is better for this procedure.
- Blend the acid required for the pH reduction in the same bucket of dissolving calcium chloride following this procedure. Chelate the calcium with SC-1000.
- Purge the pool with SC-1000 (32 fl.oz./10,000 gallons).
- Brush thoroughly.
Continue brushing and adjusting your LSI to keep it in the yellow or green for the next several days or weeks. As long as it takes until the crystals are softened enough to be brushed off. The warmer the water, the less time this takes. If you have a heater, consider bringing the water temperature up to between 75-80ºF.
Circulation is key! Circulate 24 hours a day if possible.
How to prevent calcium crystals
Prevention is as simple as winterizing the pool properly. Focus on where the LSI will be when the water temperature is at its lowest. In most cases with crystals, the water is expected to freeze at some point during the winter, so adjust the temperature in the Orenda LSI calculator down to 32ºF (0ºC) and add enough calcium hardness to allow your LSI to be green at that number. In most cases, it will be 500-600 ppm CH.
The type of pool cover you have determines how you should winterize your pool. Read this to learn more, and you can also listen to this podcast episode where we explain more: