What is ORP?
ORP stands for Oxidation-Reduction Potential. It measures how fast electrons can move through water, and therefore how effective chlorine can be.
Oxidation-Reduction Potential (ORP) is basically a speedometer for electron transfer through water. This matters because oxidation itself is the loss of electrons. The faster oxidation can occur, the more efficient and effective chlorine can be. ORP is a good measurement (in Millivolts, mV) of chlorine's efficacy.
Oxidation and Reduction
Oxidation is the loss of electrons, and reduction is the gain of electrons. The acronym "OILRIG" helps remember this (Oxidation Is Loss, Reduction Is Gain). We cover these topics in-depth in Orenda Academy: Four Pillars™. Specifically in Pillar 2. Here's a video excerpt from the program:
Electrons (e-) have a negative charge. So when an oxidizer like HOCl steals electrons from an oxidant, those negatively-charged electrons reduce the HOCl down into inert chlorides (Cl-). Chlorides alone can no longer oxidize because they cannot take on any more electrons. That's what we mean when we say "chlorine gets used up". We mean chlorine gets reduced.
This can be confusing because it sounds like the oxidant (contaminant) would be what gets reduced. The contaminant is being chemically burned out of the water. But in fact, it is chlorine that gets reduced based on its valence (electrical charge).
How ORP is measured
ORP is measured with electronic probes, also called ORP meters. ORP probes come standard on most commercial pool chemical controllers. As far as we know, there is no chemical test kit that will read ORP. It must be read electronically in millivolts (mV).
What are desired ORP levels?
Most health departments require ORP to be a minimum of 650 mV. The more, the better. Over 750 is considered good, and over 800 is considered amazing. We have seen ORP get over 900 only a few times, though it is exceptionally rare. Anything from 750 to 900 is where you want your ORP to be.
How accurate are ORP probes?
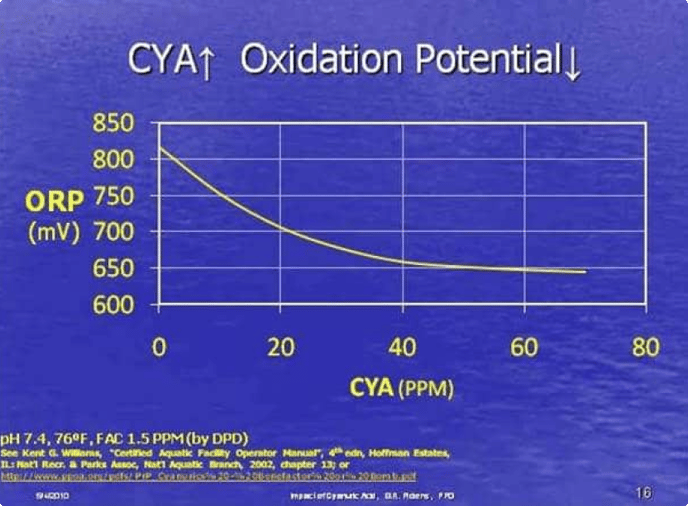
The quality and cleanliness of the probe affect its accuracy, but generally, ORP probes are pretty darn accurate. That being said, some things can interfere with their readings and "trick" ORP probes into false reasons. The main culprits of this are Cyanuric acid (CYA) and salt chlorine generators.
ORP Suppression
An odd phenomenon occurs sometimes when using salt chlorine generators (SWG). ORP can actually go down when the system is on and producing chlorine. We do not yet know 100% why this happens, but ORP probe manufacturers think it has something to do with Hydrogen gas bubbles interacting with the probe. And that seems plausible, because SWG systems do create H2 bubbles. The question is whether or not they stay in circulation long enough to reach the probes. Usually an ORP probe is before a chlorinator.
It is definitely odd, and we have it on our list of topics to research further. So if this has happened to you on your salt pool, you are not alone.
